Authors
Molly Sachdev, MD, MPH; Mina Chung, MD; Sana Al-Khatib, MD; Christopher F. Liu, MD; Lisa Miller, MS; Amit J. Shanker, MD
Correspondence
[email protected]
Summary
Boston Scientific has issued an updated medical device correction for the ACCOLADE™ family of pacemakers and cardiac resynchronization therapy pacemakers (CRT-Ps). Brady software maintenance release 6 (SMR6) is now available for affected devices. SMR6 mitigates the previously reported risk of inducing Safety Mode in an ambulatory setting due to high battery impedance and corrects unintended behaviors associated with Brady SMR5. The advisory population is also expanding to include all CRT-P and dual-chamber extended life (DR-EL) devices because of a residual risk (7.6% probability) of early device replacement related to high battery impedance–induced ZIP™ telemetry disablement. As a result, some devices may not achieve original projected longevity.
Devices Affected
The advisory population includes all serialized DR-EL pacemakers and CRT-Ps from the ACCOLADE family. In addition, ACCOLADE DR-SL and SR-SL devices with a use-by-date on or before 30 June 2025 are included. The ACCOLADE family includes ACCOLADE, PROPONENT™, ESSENTIO™, and ALTRUA™ 2 SR-SL, DR-SL, and DR-EL pacemakers, as well as VISIONIST™ and VALITUDE™ CRT-Ps. Device-specific inclusion should be confirmed using Boston Scientific’s lookup tool.
History & Current Clinical Issue
As noted in prior communications from HRS dated June 21, 2021, December 14, 2023, and December 17, 2024, Boston Scientific identified several subpopulations of pacemakers, including CRT-P devices, with an elevated risk of entering an irreversible Safety Mode during telemetry or other higher-power operations due to high battery impedance that can occur later in device life. This appears to be due to a battery cathode processing technique that resulted in a higher concentration of lithium salts, which can be associated with high battery impedance later in device life.
Safety Mode causes the device to shift to VVI 72.5 bpm, 5.0V at 1.0 ms pacing and sensing in unipolar mode. This could lead to pacing inhibition and pauses (e.g. due to myopotential inhibition), muscle stimulation (e.g. skeletal muscle or phrenic nerve stimulation), heart failure decompensation, or rapid battery depletion. Adverse events with loss of pacing can result in serious injury or life-threatening outcomes. From the Q1 2026 BSC Product Performance Report, there have been three (3) patients with pacemakers whose devices activated Safety Mode due to high battery impedance; these patients experienced syncope requiring hospitalization and later died. There has been one (1) patient whose device activated Safety Mode due to high battery impedance and died after the replacement procedure
In August, 2025, Boston Scientific launched Brady SMR5 Model 3869 v2.04 software designed to prevent potential initiation of Safety Mode due to high battery impedance conditions. The software update enabled detection of an elevated battery impedance via a telemetry activated battery test, triggering a red alert (voltage is too low for projected remaining capacity, known as Code-1003) and disabling ZIPTM telemetry (wandless two-way radiofrequency communication) when the battery reaches a high impedance state. Unfortunately, after release of the software update, some unintended behaviors were noted, as discussed in the HRS Advisory dated October 10, 2025 (Boston Scientific Accolade Pacemaker Software Update and Revision): The software disabled active telemetry but did not disable telemetry wake-ups.The software additionally had the potential to initiate a false positive response in the presence of a magnet, leading to disabling of wandless telemetry sessions and triggering on LATITUDE remote monitoring an alert message indicating an erroneous explant indicator stating that LATITUDE is no longer available and prompting providers to contact Technical Services.
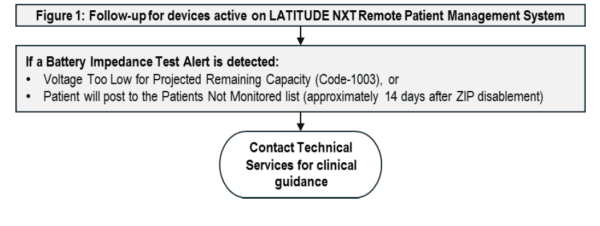
With this current software update, Boston Scientific reports that Brady SMR6 (Model 3869 v2.05) resolves incomplete ZIP disablement behavior and magnet-induced false-positive battery impedance testing. For remotely monitored patients, if ZIP becomes disabled because of high battery impedance, the device will post to the LATITUDE NXT “Patient Not Monitored” page after 14 days rather than generate a “Remote Monitoring Disabled” alert. Note that this means that if there is high battery impedance and ZIP is disabled, there is no notification and there will be a lag of 14 days before notification via LATITUDE NXT that the patient is not connected. There also remains a residual risk of in-clinic Safety Mode being induced by wanded telemetry, particularly in pacemaker-dependent patients who are not monitored on LATITUDE NXT and who have a CRT-P or DR-EL device with three years or less remaining longevity
Boston Scientific: Recommendations
Please consult statement and flow chart for more details*
- Upgrade LATITUDE™ Model 3300 programmers with Model 3869 v2.05 software (Brady SMR6).
- Upgrade pacemaker software in-clinic by interrogating the device with a programmer upgraded with Brady SMR6 (Model 3869 v2.05).
- Patients at risk of harm from Safety Mode who haven’t already received Brady SMR5: Promptly schedule an in-person follow-up if four (4) or less years of longevity time are remaining. Patients at potential risk of harm include those who are pacemaker dependent or patients with CRT-P devices who may be at risk for worsening of heart failure.
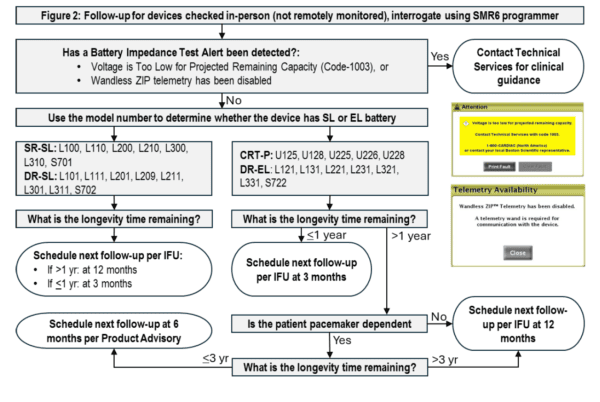
Note, the footer of the device follow-up report identifies the device firmware version. If the parenthetical at the end of the reported Firmware Version is “(3.10)” or greater, the device has been updated to either Brady SMR5 or SMR6. - All other patients: Schedule the next in-person follow up at a frequency described in the IFU: every 12 months if battery longevity exceeds one year or every 3 months if the battery status reaches One-Year-Remaining.
- Patients at risk of harm from Safety Mode who haven’t already received Brady SMR5: Promptly schedule an in-person follow-up if four (4) or less years of longevity time are remaining. Patients at potential risk of harm include those who are pacemaker dependent or patients with CRT-P devices who may be at risk for worsening of heart failure.
- Follow devices using the applicable flow chart below, based on the remote monitoring status (Figure 1 and 2).
- Update the medical record for each patient with an affected device (see Appendix A) by appending this letter to ensure continuous awareness throughout the device’s remaining service life.
Manufacturer Statement
Boston Scientific states that prophylactic replacement before confirmation of high battery impedance is not recommended. Devices experiencing ZIP disablement because of the battery impedance test require replacement. Boston Scientific further reports that single-chamber (SR) and DR standard life (SL) devices are performing within anticipated longevity expectations, but advisory devices remain unchanged for the currently recommended software upgrade.
HRS Perspective
HRS advises clinicians to confirm device status promptly, ensure software is updated, and maintain close monitoring of patients with devices under this advisory. Where remote monitoring is unavailable or incomplete, in-person follow-up should be prioritized, especially in pacemaker-dependent patients and those with limited remaining longevity. Clinicians should avoid unnecessary prophylactic replacement unless high battery impedance has been confirmed or device behavior warrants intervention. Receipt of a “Remote Monitoring Disabled” alert should prompt an in-clinic evaluation unless remote monitoring has been restored. “Remote Monitoring Disabled” alerts will not be provided for devices upgraded to SMR6 that have RF disabled due to high battery impedance (will appear on not monitored list).
Follow-up per the flow chart will require in-clinic evaluations every 3 months for all listed devices with ≤ 1 year longevity time remaining and in pacemaker-dependent patients with CRT-P or DR-EL devices every 6 months when longevity time remaining is between 1 to 3 years. Attention to high battery impedance during these clinic visits will be important, since if in-clinic telemetry triggers Safety Mode, there may be no notification for 14 days. If in clinic telemetry triggers safety mode, there will be an immediate alert on the programmer. If there is a pre-existing FC1003 or RF telemetry disablement, the screens in the flow chart will appear to alert the health care provider.
Reporting & Support
Adverse events should be reported to Boston Scientific or the FDA MedWatch program. Additional information, including a device lookup tool and product performance resources, is available through Boston Scientific Technical Services.
FDA’s MedWatch
Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report online
- Regular Mail or Fax: Download form or call 1- 800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800-FDA-0178.
Topic
- Device Therapy
- Electrophysiology
Manufacturer
- Boston Scientific
Device Type
- CRT-Ps
- Other
- Pacemakers
Related Resources

HRStv Podcast
Episode 136: ACC 2026 – The CHAMPION AF Trial
March 29, 2026

HRStv Podcast
Episode 135: ICE Image of the Month – PFA and VT Ablation (Part II)
March 20, 2026

The Lead
Podcasts
The Lead Episode 141: A Discussion of Impact of GLP-1 Receptor Agonist Therapy on Atrial Fibrillation Recurrence After Catheter Ablation in Obese Patients: A Real-World Data Analysis
March 19, 2026