May 10, 2023 – Medtronic has distributed notification regarding the potential for reduced- or no-energy output during high voltage (HV) therapy (typically 0-12J) in implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy defibrillators (CRT-Ds) (manufactured after July 2017) with a specific (glassed) feedthrough, including ICDs and CRT-Ds that are currently available for new implantation.
Authors
Molly Sachdev, MD; Amit J. Shanker, MD; Lisa Miller, MS; Anne Marie Smith, MBA; Andrew Krahn, MD; Christopher F. Liu, MD
Correspondence
[email protected]
Impacted Models
- Cobalt™ XT/Cobalt™/Crome™ ICDs and CRT-Ds
- A subset of: Claria MRI™/Amplia MRI™/Compia MRI™/Viva™/Brava™ CRT-Ds
- A subset of: Visia AF™/Visia AF MRI™/Evera™/Evera MRI™/Primo MRI™/Mirro MRI™ ICDs
Background of Recall
Medtronic is reporting 27 devices (0.003%) of approximately 816,000 devices distributed worldwide that have experienced a reduced- or no-energy HV therapy due to unintended current pathways that could form in these devices manufactured with a glassed feedthrough (after 2017). There have been no deaths due to this issue identified in the current population since 2017. Medtronic does note that a broader analysis outside of the above population (with implants dating back to 2012) identified two deaths in which there was evidence suggesting a device-related reduced- or no-energy HV therapy occurred.
In some cases, a 50% drop in all pacing lead impedances may be displayed on device check after shock therapies are delivered (whether normal or abnormal energy delivered); however, lead function is not affected. This drop in pacing lead impedance is expected to persist once it is observed.
Should this issue occur, pacing, sensing, episode detection, anti-tachycardia pacing (ATP) therapies, battery longevity, and telemetry remain functional. When devices in the glassed feedthrough population are programmed exclusively in the B>AX configuration, the estimated risk for a reduced- or no-energy HV therapy is 0.002% at five (5) years and 0.005% at nine (9) years. These estimates for device failure after reprogramming are consistent with historical data for device performance across all Medtronic manufactured ICDs, and to industry standards for device performance.
Recall Issue Summary
There is an increased potential for a reduced- or no-energy HV therapy in the AX>B configuration when all the following conditions are met:
- The device has a glassed feedthrough (manufactured after July 2017).
- There is significant separation of the layers of insulation materials in the feedthrough components of the device header.
- An unintended current pathway forms within the void created by the insulation separation, capable of conducting high levels of current during HV therapy.
When an unintended current pathway is detected during HV therapy, the Short Circuit Protection (SCP) feature may trigger. This behavior can be intermittent; both full-energy and reduced-energy HV therapies within the same episode have been observed. SCP events may also be lead-related; for both lead-related and device-related unintended current pathways, the defibrillation waveform is truncated early in the energy delivery sequence, resulting in reduced or no energy being delivered (~0-12J) – so that it may be difficult to distinguish between the two mechanisms.
Through April 10 2023, 27 devices have been identified as experiencing a reduced or no-energy HV therapy (0.003% out of 816,000 distributed devices with glassed feedthroughs). Of these, 26 were in devices with an AX>B delivered pathway (i.e., this phenomenon has been seen in the B>AX configuration but much more rarely). Based on an analysis of patients with a glassed feedthrough device and with a history of HV therapy, the observed rate for this issue is 0.03%. Medtronic’s communication provides a Table for projected rates of occurrence and risk for harm.
Potential harms related to reduced- or no-energy HV therapy include failure to terminate an arrhythmia, which could lead to death, as well as complications associated with device replacement and/or unnecessary lead replacement if the reduced- or no-energy HV therapy is erroneously attributed to a lead failure.
Manufacturer Recommendations
- Prophylactic device replacement is NOT recommended.
- Program all HV therapy pathways B>AX in all therapy zones to minimize the risk for this issue.
- Note: Using “Get Medtronic Nominals” will require manual reprogramming of Rx5 and Rx6 to B>AX for all ventricular therapies.
- or patients remotely followed via CareLink, your Medtronic representative will provide a report to assist with identifying patients who may have one or more HV therapy pathways programmed AX>B. You may contact your local representative to obtain an updated copy of the report at any time.
- Prioritize reprogramming patients who have both a history of HV therapy and Rx1 programmed AX>B.
- Rx1 provides the greatest statistical likelihood to resolve an arrhythmia, and therefore it is important to minimize risk of a reduced- or no-energy HV therapy in the first sequence.
- For remaining patients with AX>B programming in any HV therapy sequence, schedule (with appropriate discretion) the next follow-up for in-clinic reprogramming to minimize potential for reduced- or no-energy HV therapies to occur.
- Per standard practice, check tachyarrhythmia episodes to determine effectiveness of therapies that have been delivered.
- Instruct patients to contact the clinic if they receive HV therapy or hear an audible tone coming from their device.
- Verify delivered energy is consistent with programmed energy in the Episode Summary.
Note: The image below highlights an episode experiencing intermittent reduced HV therapies (red boxes).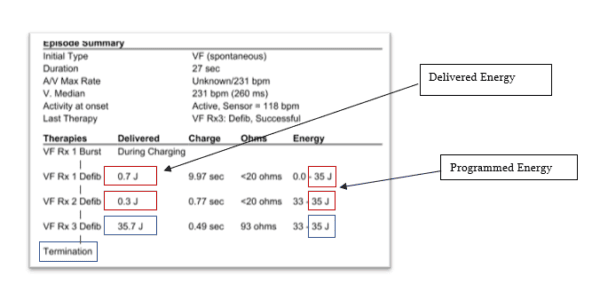
- Contact Medtronic Technical Services (1-800-929-4043) or your local representative if one of the following is observed as these may be an indication of either a device or lead-related issue:
- Reduced- or no-energy HV therapy is displayed in Episode Text (regardless of programmed pathway)
- A persistent drop of approximately 50% in all (RA, RV and LV) pacing lead impedance measurements as this may be an indication of increased potential for a future reduced- or no-energy therapy.
HRS Recommendations
HRS strongly encourages its members to read the Medtronic Safety Notification and use best clinical judgment for each individual patient. HRS agrees that prophylactic generator change is not necessary in most cases and not recommended. Medtronic currently plans to continue making these devices available for implant. Therefore, HRS recommends following these programming recommendations for all newly implanted Medtronic ICDs and CRT-Ds (also subject to this advisory).
Reporting Contact
Adverse reactions or quality problems experienced with the use of this product may be reported to your national regulatory body, including the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm.
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800- FDA-0178.
- For additional questions, contact your local Medtronic Representative or Medtronic Technical Services at 1-800-723-4636.
Topic
- Regulatory
- Regulatory Agencies
- U.S. Food and Drug Administration (FDA)
Resource Type
- Safety Alerts
Manufacturer
- Medtronic
Device Type
- ICDs & CRT-Ds
FDA Class
- Class II
Related Resources

Safety Alerts
Boston Scientific ACCOLADE™ Family Pacemakers: Brady SMR6 Software Update and Advisory Population Expansion
May 8, 2026

Safety Alerts
Device–Device Interaction Between Medtronic Sphere-9™ Ablation Catheter and Biotronik ICD/CRT-D Systems
April 16, 2026

Safety Alerts
Update to Boston Scientific RELIANCE G Defibrillation Leads Connected to Non-Boston Scientific ICD Generators
February 3, 2026